 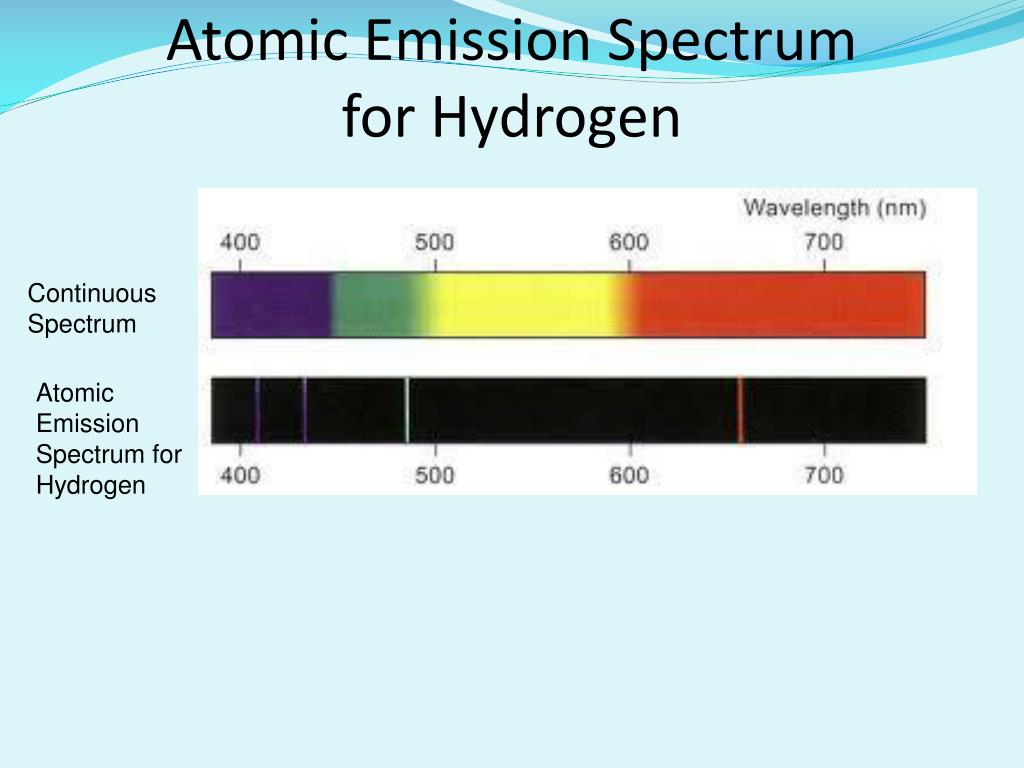
Ozone (O 3) and molecular oxygen (O 2) absorb light with wavelengths under 300 nm, meaning that X-ray and ultraviolet spectroscopy require the use of a satellite telescope or rocket mounted detectors. While all spectroscopy looks at specific bands of the spectrum, different methods are required to acquire the signal depending on the frequency. The atmosphere blocks some wavelengths but it is mostly transparent for visible light and a wide range of radio waves.Īstronomical spectroscopy is used to measure three major bands of radiation in the electromagnetic spectrum: visible light, radio waves, and X-rays. When white light is passed through unexcited atomic hydrogen and then through a slit and prism, the transmitted light is lacking in intensity at the same wavelengths as are emitted spectra The recorded absorption spectrum is also a line spectrum and the photographic negative of the emission spectrum.Opacity of the Earth's atmosphere for different wavelengths of electromagnetic radiation. The light emitted as these atoms fall to lower energy states is responsible for the spectrum Absorption Spectra Although a single atom can be in only one excited state at a time, the collection of atoms contains all possible excited states. Any sample of reasonable size contains an enormous number of atoms. Thus anĮmission spectrum, which is a photographic recording of the separated wavelengths is called as line spectrum. The light emitted by a sample of excited hydrogen atoms (or any other element) can be passed through a prism and separated into certain discrete wavelengths. The characteristic lines in atomic spectra can be used in chemical analysis to identify unknown atoms in the same way as fingerprints are used to identify people Exampleīoth of the Emission And absorption spectrum is described in below figure Emission Spectra Advertisements Such spectra are called line spectra or atomic spectraĮvery element has a unique line emission spectrum. The emission spectra of atoms in the gas phase, on the other hand, do not show a continuous spread of wavelength from red to violet, rather they emit light only at specific wavelengths with dark spaces between them. The spectrum of the visible light, is continuous as all wavelengths (red to violet) of the visible light are represented in the spectra. The study of emission or absorption spectra is referred as spectroscopy Line Spectrum The wavelengths which are absorbed are missing and come as dark lines.Īn absorption spectrum is like the photographic negative of an emission spectrum Spectroscopy The sample absorbs radiation of certain wavelengths. Absorption SpectrumĪbsorption spectrum is the spectrum obtained when radiation is passed through a sample of material. 
Sample gives up the absorbed energy, is recorded. It is noticed when radiations emitted from source are passed through a prism & received on photographic plate.Įmission spectrum is produced by supplying energy to a sample by heating it or irradiating it and the wavelength (or frequency) of the radiation emitted, as the The spectrum of radiation emitted by a substance that has absorbed energy is called an emission spectrum. Hence, this spectrum known as continuous spectrum. These colors are so continuous that each of them merges into the next. When white light is analyzed by passing through prism, it splits into seven colors. VIBGYOR (V) = Shortest wavelength 400nm R = Longest wavelength 450 nm Continuous spectrum When white light is passed through prism, it splits into band of seven colors called spectrum. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
